
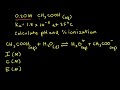
Acetic Acid Equilibrium Concepts
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the initial concentration of acetic acid in the solution?
0.30 M
0.10 M
0.20 M
0.40 M
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the Ka value of acetic acid used in this problem?
1.8 x 10^-6
1.8 x 10^-3
1.8 x 10^-4
1.8 x 10^-5
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the Ka value being less than one?
It indicates a strong acid
It indicates a weak acid
It indicates a neutral solution
It indicates a basic solution
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In an ICE table, what does the 'C' stand for?
Concentration
Change
Constant
Coefficient
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the equilibrium concentration of hydronium ions represented by in the ICE table?
0.20 - x
0.20 + x
2x
x
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of using an approximation in this calculation?
To avoid using a quadratic equation
To simplify the balanced equation
To change the initial concentration
To increase the accuracy of the result
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why can we assume that 0.20 - x is approximately equal to 0.20?
Because x is a very small number
Because the temperature is high
Because acetic acid is a strong acid
Because x is a large number
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
