
Spectrophotometry and Linear Regression Concepts
Interactive Video
•
Chemistry, Mathematics, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the absorbance value of the potassium permanganate solution measured at 540 nm?
0.53
0.25
0.35
0.75
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of using a spectrophotometer in this experiment?
To determine the absorbance of the solution
To measure the weight of the solution
To find the temperature of the solution
To calculate the volume of the solution
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the wavelength 540 nm in this experiment?
It is the wavelength at which the solution is least reactive
It is the wavelength at which the solution has maximum absorbance
It is the wavelength at which the solution is most stable
It is the wavelength at which the solution changes color
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the relationship between absorbance and concentration in a solution?
Logarithmic
Exponential
Linear
Quadratic
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What tool did the narrator use to perform linear regression?
Excel
MATLAB
Python
Desmos
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
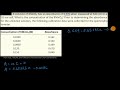
What is the slope (m) in the linear equation obtained from the regression?
1.23456
0.0086
3.14159
5.65333
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the y-intercept (b) in the linear equation obtained from the regression?
1.0000
0.1234
5.65333
0.0086
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
