
Electrophilic and Nucleophilic Aromatic Substitution
Interactive Video
•
Chemistry, Science
•
10th Grade - University
•
Practice Problem
•
Hard
Amelia Wright
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
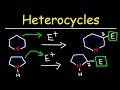
Why does bromination of pyrrole not require a Lewis acid catalyst?
Pyrrole is less reactive than benzene.
Pyrrole is not an aromatic compound.
Pyrrole is more nucleophilic than benzene.
Bromine is a weak electrophile.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which position does bromine prefer to substitute in pyrrole?
Position 4
Position 3
Position 2
Position 1
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is more nucleophilic than benzene?
Furan
All of the above
Thiophene
Pyrrole
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is thiophene less nucleophilic than furan?
Furan has a larger ring size.
Thiophene is not aromatic.
Sulfur's 3p orbital does not overlap effectively with carbon's 2p orbital.
Sulfur is more electronegative than oxygen.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does electrophilic attack prefer the two position in pyrrole?
It allows for more resonance structures.
It is less sterically hindered.
It is closer to the nitrogen atom.
It is more electronegative.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In pyridine, which position is favored for electrophilic substitution?
Position 2
Position 3
Position 1
Position 4
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is pyridine less reactive than benzene in electrophilic aromatic substitution?
Pyridine is not aromatic.
Pyridine has a nitrogen atom that deactivates the ring.
Pyridine is more nucleophilic.
Pyridine has a larger ring size.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
