
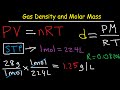
Gas Laws and Molar Mass Concepts
Interactive Video
•
Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of nitrogen gas (N2)?
44.01 g/mol
28.02 g/mol
14.01 g/mol
32.00 g/mol
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
At STP, what volume does one mole of gas occupy?
20.0 liters
24.0 liters
22.4 liters
18.0 liters
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which equation is used to derive the formula for gas density?
Ideal Gas Law
Charles's Law
Boyle's Law
Avogadro's Law
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formula for density derived from the ideal gas law?
Density = Pressure / (R * Temperature)
Density = Molar Mass / Volume
Density = (Pressure * Molar Mass) / (R * Temperature)
Density = (R * Temperature) / Pressure
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you convert pressure from torr to atm?
Multiply by 760
Divide by 760
Multiply by 101.3
Divide by 101.3
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the density of nitrogen gas at 25°C and 872 torr?
1.25 g/L
1.47 g/L
1.31 g/L
1.50 g/L
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of a gas if a 14.5 g sample occupies 3 liters at 300 K and 650 mmHg?
120.5 g/mol
150.0 g/mol
139.1 g/mol
130.2 g/mol
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
