
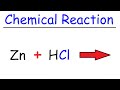
Zinc and Hydrochloric Acid Reactions
Interactive Video
•
Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Easy
Lucas Foster
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of reaction occurs between zinc metal and hydrochloric acid?
Single replacement reaction
Decomposition reaction
Synthesis reaction
Double replacement reaction
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of a zinc ion in the reaction with hydrochloric acid?
1-
1+
2-
2+
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What gas is produced when zinc reacts with hydrochloric acid?
Oxygen
Nitrogen
Chlorine
Hydrogen
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the reaction, what happens to zinc?
It forms a precipitate
It remains unchanged
It is oxidized
It is reduced
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following metals cannot displace hydrogen from hydrochloric acid?
Zinc
Copper
Magnesium
Aluminum
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of consulting the activity series of metals?
To measure the temperature change in reactions
To determine the solubility of metals
To predict the color change in reactions
To check if a metal can displace another in a reaction
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in balancing the chemical equation for the reaction?
Balance the oxygen atoms
Balance the chloride ions
Balance the zinc atoms
Balance the hydrogen atoms
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
