
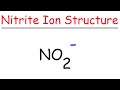
Understanding the Lewis Structure of the Nitrite Ion
Interactive Video
•
Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total number of valence electrons in the nitrite ion?
19
18
17
16
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many lone pairs are on the central nitrogen atom in the nitrite ion?
Three
Four
Two
One
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the maximum number of electrons that nitrogen can have around it according to the oxide rule?
10
8
12
6
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which type of bond is formed between nitrogen and one of the oxygen atoms in the nitrite ion?
Triple bond
Double bond
Quadruple bond
Single bond
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the double bond in the resonance structure of the nitrite ion?
It becomes a triple bond
It moves to the other oxygen atom
It disappears
It remains unchanged
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molecular geometry around the central nitrogen atom in the nitrite ion?
Tetrahedral
Linear
Bent
Trigonal planar
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the hybridization of the nitrogen atom in the nitrite ion?
sp3d
sp3
sp2
sp
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
