
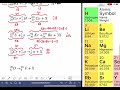
Balancing Nuclear Reactions Through Mass and Atomic Numbers
Interactive Video
•
Physics, Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary method for balancing nuclear reactions?
By the number of neutrons
By the number of atoms
By the number of electrons
By mass number and atomic number
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a nuclear reaction, if the left side has a mass number of 27, what should the right side have?
A mass number of 26
A mass number of 28
A mass number of 25
A mass number of 27
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which element is represented by atomic number 11 in the periodic table?
Hydrogen
Iron
Sodium
Calcium
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the mass of each X particle if 3X has a total mass of 3?
3
2
1
0
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What particle has a mass number of 1 but no protons?
Alpha particle
Neutron
Electron
Proton
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is another name for an electron in nuclear reactions?
Neutron
Beta particle
Alpha particle
Gamma ray
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If an element has an atomic number of 20, what is it?
Iron
Calcium
Sodium
Helium
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
