
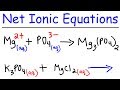
Writing Net Ionic Equations for Double Replacement Reactions
Interactive Video
•
Chemistry, Science, Physics
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in writing a net ionic equation?
Predicting the products
Balancing the equation
Identifying spectator ions
Writing the molecular equation
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following compounds is always soluble?
Silver chloride
Lead iodide
Sodium nitrate
Magnesium phosphate
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the lead ion in lead nitrate?
+3
+2
+1
+4
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of balancing a molecular equation?
To predict the products
To identify spectator ions
To ensure mass conservation
To determine solubility
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which ions are considered spectator ions in the reaction of sodium iodide and lead nitrate?
Lead and iodide ions
Lead and nitrate ions
Sodium and nitrate ions
Sodium and iodide ions
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the net ionic equation for the reaction between sodium iodide and lead nitrate?
2Na+ + 2NO3- → 2NaNO3
2I- + Pb2+ → PbI2
2Na+ + 2I- + Pb2+ + 2NO3- → PbI2 + 2Na+ + 2NO3-
NaI + Pb(NO3)2 → PbI2 + 2NaNO3
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of reaction occurs when two aqueous solutions form a solid product?
Decomposition reaction
Precipitation reaction
Synthesis reaction
Combustion reaction
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
