
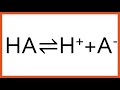
The Role and Function of Buffer Solutions in Chemical Reactions
Interactive Video
•
Chemistry, Science, Biology
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the color of a universal indicator when an acid is added to a neutral solution?
It turns blue
It turns red
It turns yellow
It remains green
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary function of a buffer solution?
To increase the pH of a solution
To decrease the pH of a solution
To change the color of a solution
To maintain a stable pH despite the addition of acids or alkalis
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does HA represent in a buffer solution?
A strong base
A weak acid
A strong acid
A neutral compound
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a buffer solution, what is the role of the weak acid HA when exposed to OH-?
It forms a strong acid
It remains unchanged
It forms water and a conjugate base
It forms a strong base
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens if an excessive amount of OH- is added to a buffer solution?
The buffer solution becomes more acidic
The buffer solution becomes more alkaline
The buffer solution remains unchanged
The buffer solution evaporates
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is a soluble salt added to a buffer solution?
To increase the acidity
To change the color of the solution
To decrease the alkalinity
To provide additional A- ions
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the result of the reaction between A- and H+ in a buffer solution?
No reaction occurs
Formation of a strong base
Formation of a weak acid
Formation of a strong acid
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
