
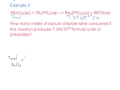
Stoichiometry Concepts and Calculations
Interactive Video
•
Chemistry, Science, Mathematics
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary focus of lesson 9.3 in stoichiometry?
Balancing chemical equations
Calculating molar mass
Understanding mixed inputs and outputs
Identifying reaction types
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is NOT a typical output in stoichiometry problems?
Volume
Number of particles
Color change
Mass
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which tool is essential for solving stoichiometry problems?
A ruler
A thermometer
A microscope
A periodic table
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of using a mole map in stoichiometry problems?
To balance equations
To measure temperature
To identify reaction types
To convert between different units
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the calcium carbonate reaction example, what is the first step after writing the chemical equation?
Measuring the temperature
Balancing the equation
Calculating the molar mass
Identifying the precipitate
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many liters of CO2 are produced from 12.3 grams of calcium carbonate at STP?
1.5 liters
3.0 liters
4.0 liters
2.75 liters
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the barium chloride reaction, what is the precipitate formed?
Barium sulfate
Calcium chloride
Sodium chloride
Barium phosphate
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
