
Chemical Composition and Formulas
Interactive Video
•
Chemistry, Science, Biology
•
6th - 8th Grade
•
Practice Problem
•
Easy
Patricia Brown
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following atoms are considered reactive due to their valence electrons?
Argon and Krypton
Carbon and Nitrogen
Oxygen and Hydrogen
Helium and Neon
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the chemical formula H2O represent?
Two atoms of oxygen and one atom of hydrogen
Two atoms of hydrogen and one atom of oxygen
One atom of hydrogen and two atoms of oxygen
Two atoms of hydrogen and two atoms of oxygen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the chemical formula CO2, what does the subscript '2' indicate?
Two atoms of oxygen
Two molecules of oxygen
Two atoms of carbon
Two molecules of carbon
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
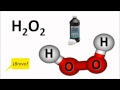
What is the chemical formula for hydrogen peroxide?
HO2
H2O
H2O3
H2O2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many hydrogen atoms are in a molecule of sulfuric acid (H2SO4)?
Two
Three
One
Four
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does a coefficient in a chemical equation represent?
The number of atoms in a molecule
The number of molecules involved in a reaction
The number of electrons in an atom
The number of elements in a compound
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If you have 4 molecules of NaCl, what is the correct way to write this using a coefficient?
NaCl
Na4Cl
4NaCl
NaCl4
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
