
Supersaturated Solutions and Solubility Concepts
Interactive Video
•
Chemistry, Science, Biology
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary focus of the video tutorial?
Periodic table
Atomic structure
Solutions and saturation
Chemical reactions
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is necessary for solute and solvent molecules to interact?
Low temperature
Presence of a catalyst
Breaking up of solute and solvent
High pressure
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What drives the formation of solutions?
Increase in pressure
Decrease in temperature
Increase in entropy
Decrease in entropy
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
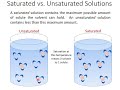
What defines a saturated solution?
Contains more solute than the solvent can hold
Contains only solvent
Contains the maximum solute the solvent can hold
Contains no solute
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does temperature affect solubility for solid solutes?
Solubility decreases with increasing temperature
Solubility increases with increasing temperature
Solubility remains constant with temperature
Temperature has no effect on solubility
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when a hot saturated solution is cooled carefully?
It becomes supersaturated
It becomes unsaturated
It remains saturated
It evaporates
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a key characteristic of supersaturated solutions?
They are stable
They are unstable
They have no solute
They are always cold
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
