
Chemical Reaction Energy Concepts
Interactive Video
•
Chemistry, Science, Physics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the minimum energy required for molecules to collide and react called?
Thermal Energy
Potential Energy
Activation Energy
Kinetic Energy
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
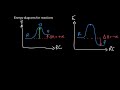
In a reaction coordinate diagram, what does the peak represent?
Transition State
Products
Equilibrium
Reactants
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the energy of the system when products have higher energy than reactants?
The reaction stops
The reaction is endothermic
The reaction is exothermic
The reaction reaches equilibrium
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the sign of delta H for an exothermic reaction?
Undefined
Zero
Negative
Positive
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
During the reaction of a fluoride ion with a methyl chloride molecule, what happens to the carbon-chlorine bond?
It shortens and strengthens
It remains unchanged
It lengthens and weakens
It strengthens
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the configuration of maximum energy in a chemical reaction called?
Reactant State
Product State
Transition State
Intermediate State
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of the transition state in a chemical reaction?
It is the point where energy is minimum
It is the end point of the reaction
It is the point of no return with maximum energy
It is the starting point of the reaction
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
