
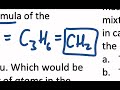
Mole Concepts and Empirical Formulas
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the relationship between the mass of a substance and its moles?
Mass is directly proportional to moles.
Mass is inversely proportional to moles.
Mass has no relation to moles.
Mass is equal to moles.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many particles are there in one mole of a substance?
6.02 x 10^25
6.02 x 10^23
6.02 x 10^22
6.02 x 10^24
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the percent composition of carbon in methane (CH4)?
25%
100%
75%
50%
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you determine the empirical formula from percent composition?
Use the periodic table to find the empirical formula.
Convert percentages to moles directly.
Convert percentages to grams, then to moles, and find the simplest ratio.
Empirical formulas cannot be determined from percent composition.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a balanced reaction, what does a mole ratio represent?
The ratio of densities of reactants and products.
The ratio of masses of reactants and products.
The ratio of moles of reactants and products.
The ratio of volumes of reactants and products.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the empirical formula of a compound with 36 grams of carbon and 6 grams of hydrogen?
C3H6
CH2
C2H4
C6H12
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is necessary to find the number of atoms in a sample of copper?
The volume of copper.
The molar mass of copper.
The density of copper.
The color of copper.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
