
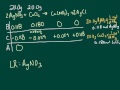
Stoichiometry Concepts and Calculations
Interactive Video
•
Chemistry, Science, Mathematics
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary objective of using a BCA table in stoichiometry?
To find the theoretical amounts of products and excess reactants
To determine the speed of a reaction
To calculate the pH of a solution
To measure the temperature change in a reaction
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the given problem, which two compounds are initially reacted?
Silver nitrate and sodium chloride
Calcium nitrate and silver chloride
Silver nitrate and calcium chloride
Calcium chloride and sodium nitrate
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you convert grams to moles in stoichiometry?
By subtracting the molar mass from the grams
By adding the grams to the molar mass
By multiplying the grams by the molar mass
By dividing the grams by the molar mass
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the term 'limiting reactant' refer to?
The reactant that changes color during the reaction
The reactant that speeds up the reaction
The reactant that determines the amount of product formed
The reactant that is left over after the reaction
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the limiting reactant determined in a chemical reaction?
By comparing the initial masses of reactants
By comparing the mole ratios of reactants
By observing the color change
By measuring the temperature change
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the excess reactant in a reaction?
It speeds up the reaction
It is completely used up in the reaction
It remains after the reaction is complete
It changes the color of the solution
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you convert moles of a substance to grams?
Multiply the moles by the molar mass
Divide the moles by the molar mass
Add the moles to the molar mass
Subtract the molar mass from the moles
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
