
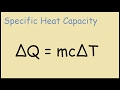
Heating Water and Energy Calculations
Interactive Video
•
Physics, Science, Mathematics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the initial temperature of the water in the hot water tank?
30°C
40°C
20°C
10°C
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which variable in the specific heat equation represents the mass of the substance?
C
Delta T
m
Delta Q
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the mass of the water calculated from its volume?
Multiply by 2
Divide by 2
Use the density of water
Use the specific heat capacity
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the specific heat capacity of water used in the calculation?
3,000 J/kg K
4,200 J/kg K
6,000 J/kg K
5,000 J/kg K
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the change in temperature (Delta T) for the water in this problem?
50°C
60°C
40°C
30°C
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How much energy is required to raise the temperature of the water to 70°C?
18,350,000 J
28,350,000 J
38,350,000 J
48,350,000 J
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formula for calculating power in terms of energy and time?
Power = Time / Energy
Power = Energy + Time
Power = Energy x Time
Power = Energy / Time
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
