
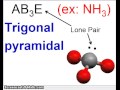
Molecular Shapes and VSEPR Theory
Interactive Video
•
Chemistry, Science, Biology
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the VSEPR theory primarily explain?
The three-dimensional arrangement of atoms in a molecule
The color of molecules
The weight of molecules
The speed of chemical reactions
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is an example of a linear molecule?
H2O
NH3
CO2
CH4
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a trigonal planar shape, how are the atoms arranged?
In a straight line
In a square
In a pyramid
In a triangle on the same plane
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main difference between trigonal planar and trigonal pyramidal shapes?
The color of the molecule
The number of atoms involved
The presence of a lone pair of electrons
The type of atoms involved
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which shape is most commonly associated with four atoms surrounding a central atom?
Bent
Tetrahedral
Trigonal planar
Linear
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many faces does a tetrahedron have?
Five
Six
Four
Three
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What causes the bent shape in molecules like H2O?
The temperature of the environment
The type of atoms involved
The presence of lone pairs of electrons
The presence of double bonds
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
