
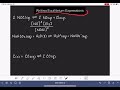
Equilibrium Expressions and KC Values
Interactive Video
•
Chemistry, Science, Mathematics
•
10th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the difference between K and Q in equilibrium expressions?
K is for reactants, while Q is for products.
K is used for concentrations at equilibrium, while Q is for any concentrations.
K is for gases only, while Q is for liquids.
K is a constant, while Q varies with temperature.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the KC value calculated in equilibrium expressions?
By using the pressure of gases.
By using the temperature of the reaction.
By using the molarity of reactants and products.
By using the volume of the container.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of stoichiometric coefficients in equilibrium expressions?
They are ignored in the expression.
They are subtracted from the expression.
They are used as exponents in the expression.
They are added to the expression.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why are pure liquids excluded from equilibrium expressions?
They are always in excess.
They have a constant concentration.
They do not have a defined molarity.
They react too quickly.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why can't pure solids be included in equilibrium expressions?
They have a fixed volume.
They do not dissolve in water.
They do not have a defined molarity.
They are always reactants.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the equilibrium expression if the chemical equation is reversed?
The expression is inverted.
The expression is doubled.
The expression is halved.
The expression remains the same.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does changing the stoichiometry of a reaction affect the KC value?
It only affects the Q value.
It changes the KC value.
It only affects the reactants.
It does not affect the KC value.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
