
Understanding pH Measurement Concepts
Interactive Video
•
Chemistry, Science, Physics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
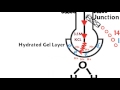
What is the primary function of the glass bulb in a pH electrode?
To conduct electricity
To come in contact with the solution being tested
To store the electrolyte solution
To house the internal microchip
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the typical concentration of the KCl solution used in pH electrodes?
1.0 molar
4.0 molar
2.5 molar
3.5 molar
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which type of junction is typically used for high concentration solutions like plant nutrient solutions?
Ceramic Junction
Cloth Junction
Metal Junction
Open Junction
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important for the gel layer on the glass bulb to be hydrated?
To keep the electrolyte solution from leaking
To ensure accurate pH measurement
To maintain the internal microchip
To prevent the glass bulb from breaking
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does pH stand for?
Potential of Hydrogen
Power of Hydroxide
Pressure of Helium
Presence of Heat
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the number of hydrogen ions affect the pH value of a solution?
Fewer hydrogen ions result in a lower pH
More hydrogen ions result in a higher pH
More hydrogen ions result in a lower pH
The number of hydrogen ions does not affect pH
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of the electrical charge in determining the pH value?
It influences the migration of hydrogen ions
It powers the internal microchip
It prevents the leakage of the electrolyte solution
It stabilizes the glass bulb
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
