
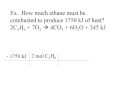
Chemical Reactions and Energy Changes
Interactive Video
•
Chemistry, Science, Physics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are the two main types of chemical reactions based on energy changes?
Endothermic and Exothermic
Combustion and Synthesis
Decomposition and Displacement
Oxidation and Reduction
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In which unit is energy most commonly measured in chemical reactions?
Kilojoules
Joules
Watts
Calories
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the energy of products compared to reactants in an endothermic reaction?
Energy is released
Energy remains the same
Products have more energy
Products have less energy
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the sign of Delta H in an endothermic reaction?
Positive
Undefined
Negative
Zero
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In an exothermic reaction, how does the energy of the products compare to the reactants?
Energy remains constant
Energy is absorbed
Products have less energy
Products have more energy
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the sign of Delta H in an exothermic reaction?
Negative
Zero
Positive
Undefined
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many moles of ethane are needed to produce 1750 kilojoules of heat?
4.89 moles
2.54 moles
3.21 moles
5.07 moles
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
