
Thermochemistry and Calorimetry Concepts
Interactive Video
•
Chemistry, Physics, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
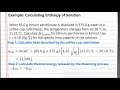
What is the initial temperature of the water in the calorimeter?
25.00 degrees Celsius
20.18 degrees Celsius
31.21 degrees Celsius
15.00 degrees Celsius
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of using a coffee-cup calorimeter in this experiment?
To find the density of lithium perchlorate
To determine the boiling point of water
To calculate the enthalpy of solution
To measure the mass of the solution
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total mass of the solution in the calorimeter?
85.0 grams
375.0 grams
500.0 grams
460.0 grams
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How much heat is absorbed by the calorimeter's contents?
21,208 joules
15,000 joules
30,000 joules
10,000 joules
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the dissolving process considered exothermic?
Because it absorbs heat from the surroundings
Because it requires energy input to dissolve
Because it releases thermal energy, increasing the temperature
Because it decreases the temperature of the solution
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the sign of the energy released by the dissolving process?
Negative
Positive
Undefined
Zero
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the heat released per 85.0 grams of lithium perchlorate?
15,000 joules
21,208 joules
-15,000 joules
-21,208 joules
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
