
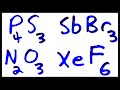
Molecular Compounds and Chemical Formulas
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main focus of the video tutorial?
Balancing chemical equations
Understanding atomic structure
Writing formulas for molecular compounds with two non-metals
Writing formulas for ionic compounds
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you express the names of elements in a molecular compound?
As electron configurations
As atomic numbers
As chemical symbols
As molecular weights
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What prefix is used for the number three in molecular compounds?
Tetra
Penta
Hexa
Tri
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the absence of a prefix before the first element in a compound name indicate?
The element is a metal
There is one atom of the element
The element is a noble gas
There are two atoms of the element
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the chemical formula for dinitrogen trioxide?
N3O2
NO2
NO3
N2O3
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the chemical symbol for antimony?
At
Am
Sb
An
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many bromine atoms are in antimony tribromide?
Three
Four
One
Two
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
