
Titration Concepts and Calculations
Interactive Video
•
Chemistry
•
11th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the initial concentration of acetic acid used in the titration experiment?
0.5 M
1.0 M
2.0 M
1.5 M
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What volume of NaOH is added to reach the equivalence point?
20 mL
25 mL
30 mL
35 mL
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
After the equivalence point, what type of problem is the titration considered?
Equilibrium problem
Stoichiometric problem
Kinetic problem
Thermodynamic problem
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
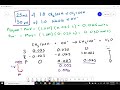
In the surf table, what is identified as the limiting reagent after the equivalence point?
Strong base (OH-)
Water
Weak acid
Acetate
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the final concentration of OH- after the equivalence point?
0.1101 M
0.0909 M
0.0707 M
0.0505 M
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the pH calculated from the pOH in this titration?
pH = pOH + 14
pH = pOH - 7
pH = 14 - pOH
pH = 7 - pOH
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the pH of the solution after adding 30 mL of NaOH?
12.9
12.5
12.0
11.5
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
