
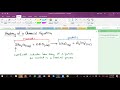
Chemical Equations and Symbols
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary purpose of using chemical equations?
To describe the physical appearance of substances
To symbolically represent chemical and physical changes
To calculate the speed of reactions
To measure the temperature of reactions
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the example given, what is formed when sodium carbonate reacts with aluminum chloride?
Table salt and aluminum carbonate
Water and carbon dioxide
Aluminum oxide
Sodium bicarbonate
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What do coefficients in a chemical equation indicate?
The type of reaction
The number of particles involved
The color of the reactants
The temperature of the reaction
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of subscripts in chemical formulas?
To describe the color of the compound
To represent the speed of a reaction
To show the number of atoms or ions in a formula unit
To indicate the state of matter
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the state symbol 'aq' represent in a chemical equation?
Solid
Liquid
Gas
Aqueous, meaning dissolved in water
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which state symbol indicates a substance is in a gaseous state?
l
s
g
aq
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many aluminum atoms are present in one formula unit of aluminum carbonate, Al2(CO3)3?
One
Two
Four
Three
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
