
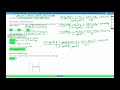
Chemical Reactions and Enthalpy Calculations
Interactive Video
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main objective of the problem presented in the video?
To balance a chemical equation
To find the atomic mass of selenium
To calculate the density of hydrogen
To determine the limiting reactant and enthalpy change
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of H2 used in the calculations?
78.97 g/mol
2.02 g/mol
1.01 g/mol
631.76 g/mol
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many moles of H2 are present in 27.3 grams?
2.06 moles
1.69 moles
8 moles
13.5 moles
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which reactant is in excess according to the video?
Selenium (Se8)
Neither is in excess
Hydrogen (H2)
Both are in excess
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the limiting reactant in the reaction?
Neither is limiting
Selenium (Se8)
Hydrogen (H2)
Both are limiting
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many moles of Se8 are required to fully react with 13.5 moles of H2?
13.5 moles
1.69 moles
2.06 moles
8 moles
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the enthalpy change for the reaction when 13.5 moles of H2 react?
42 kJ
13.5 kJ
8 kJ
238 kJ
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
