
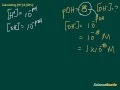
Understanding Ion Concentrations and Equations
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary focus of this video tutorial?
Identifying chemical reactions
Balancing chemical equations
Understanding ion concentrations
Calculating molar mass
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which equation is used to find the hydrogen ion concentration?
Hydrogen ion concentration = 10^pH
Hydrogen ion concentration = 10^-pH
Hydroxide ion concentration = 10^-pOH
Hydroxide ion concentration = 10^pOH
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If the pH is 2, what is the hydrogen ion concentration?
1 * 10^-1 M
1 * 10^-2 M
1 * 10^-4 M
1 * 10^-3 M
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is a pH of 4 expressed in terms of hydrogen ion concentration?
0.1 M
0.0001 M
0.001 M
0.01 M
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which equation is used to find the hydroxide ion concentration?
Hydroxide ion concentration = 10^-pOH
Hydroxide ion concentration = 10^pH
Hydroxide ion concentration = 10^-pH
Hydroxide ion concentration = 10^pOH
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
For a pOH of 3, what is the hydroxide ion concentration?
1 * 10^-3 M
1 * 10^2 M
1 * 10^3 M
1 * 10^-2 M
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the hydroxide ion concentration for a pOH of 8?
1 * 10^-6 M
1 * 10^-5 M
1 * 10^-7 M
1 * 10^-8 M
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

8 questions
Menulis rumus kimia sederhana
Interactive video
•
10th Grade

6 questions
DFM rises 1.81 percent
Interactive video
•
9th - 10th Grade

6 questions
CLEAN : Women encouraged to try firefighting as a career
Interactive video
•
9th - 10th Grade

6 questions
CLEAN : Football: Ibra nets four as PSG seal French title in record time
Interactive video
•
9th - 10th Grade

6 questions
Hearse blocks road near Trafalgar Square as part of Extinction Rebellion protests
Interactive video
•
9th - 12th Grade

2 questions
GCSE Biology - Control of Blood Glucose Concentration #56
Interactive video
•
9th - 10th Grade

6 questions
GCSE Secondary Maths Age 13-17 - Algebra: Algebra - Simplifying - Explained
Interactive video
•
10th - 12th Grade

6 questions
GCSE Secondary Maths Age 13-17 - Algebra: Algebra - Explained
Interactive video
•
10th - 12th Grade
Popular Resources on Wayground

8 questions
Spartan Way - Classroom Responsible
Quiz
•
9th - 12th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

3 questions
Integrity and Your Health
Lesson
•
6th - 8th Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

9 questions
FOREST Perception
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade
Discover more resources for Chemistry

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

13 questions
Solubility Curves
Quiz
•
10th Grade

20 questions
momentum and impulse
Quiz
•
9th - 12th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade

35 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

15 questions
Ionic Bonding
Quiz
•
10th - 11th Grade