
Properties and Behavior of Water
Interactive Video
•
Biology
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What percentage of the Earth's surface is covered by water?
90%
34%
50%
75%
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is water essential for cellular function?
It acts as a waste product.
It is a source of vitamins.
It maintains cellular shape and participates in chemical reactions.
It provides energy to cells.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
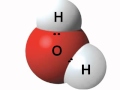
What is the shape of a water molecule?
Square
V-shaped
Tetrahedral
Linear
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which atom in a water molecule is more electronegative?
Hydrogen
Both are equally electronegative
Oxygen
Neither
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the electrons in a water molecule due to electronegativity?
They are equally shared between hydrogen and oxygen.
They spend more time around the hydrogen atoms.
They are lost to the environment.
They spend more time around the oxygen atom.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What causes a water molecule to be polar?
Equal sharing of electrons
Unequal sharing of electrons
Presence of ionic bonds
Absence of hydrogen bonds
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bond forms between water molecules?
Covalent bond
Ionic bond
Metallic bond
Hydrogen bond
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
