
Balancing Redox Reactions Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are the two main approaches to balancing redox reactions?
Balancing by temperature and pressure
Balancing by volume and density
Balancing by mass or moles and by charge
Balancing by color and texture
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When balancing by mass or moles, what is the first step?
Assigning oxidation numbers
Measuring the temperature
Adding water molecules
Heating the reaction
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it necessary to adjust coefficients when balancing equations?
To change the color of the solution
To increase the reaction speed
To ensure the reaction is exothermic
To balance the number of atoms on both sides
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What must be consistent when breaking a balanced equation into half-reactions?
The color of the reactants
The coefficients
The pressure
The temperature
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
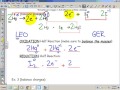
In a redox reaction, what does LEO stand for?
Large Electron Orbit
Low Energy Output
Light Emission Observation
Lose Electron Oxidation
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the GER acronym in redox reactions?
Gain Electron Reduction
General Energy Release
Gravitational Energy Ratio
Gas Emission Rate
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main focus when balancing redox reactions by charge?
Balancing the temperature
Balancing the color
Balancing the charges
Balancing the pressure
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
