
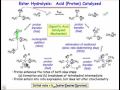
Ester Hydrolysis Reaction Mechanisms
Interactive Video
•
Chemistry
•
11th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary focus of comparing uncatalyzed and catalyzed reactions in the context of ester hydrolysis?
To determine the final products of the reaction
To understand how the catalyst appears in the rate law
To calculate the energy released in the reaction
To measure the temperature change during the reaction
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the uncatalyzed ester hydrolysis reaction, what is the role of the nucleophile?
To add to a polarized pi bond
To act as a leaving group
To stabilize the intermediate
To donate a proton
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which step is considered the rate-determining step in the uncatalyzed ester hydrolysis reaction?
Nucleophile addition
Proton transfer
Beta elimination
Formation of the carboxylic acid
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the concentration of the tetrahedral intermediate accounted for in the rate law derivation?
By assuming it is constant
By ignoring it completely
By using fast equilibrium assumptions
By direct measurement
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What assumption is made to simplify the reaction and focus on the forward rate?
The reaction is at equilibrium
The reaction is exothermic
The backward reaction is negligible
The catalyst is consumed
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the proton-catalyzed mechanism, what role does the proton play in the reaction?
It stabilizes the final product
It is consumed and not regenerated
It acts as a nucleophile
It enhances the electrophilicity of the carbonyl
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the proton affect the rate-determining step in the catalyzed reaction?
It has no effect
It accelerates the beta elimination
It changes the reaction pathway
It slows down the reaction
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Popular Resources on Wayground

8 questions
Spartan Way - Classroom Responsible
Quiz
•
9th - 12th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

3 questions
Integrity and Your Health
Lesson
•
6th - 8th Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

9 questions
FOREST Perception
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
momentum and impulse
Quiz
•
9th - 12th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade

20 questions
Naming Covalent Compounds
Quiz
•
11th Grade

35 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

15 questions
Ionic Bonding
Quiz
•
10th - 11th Grade

12 questions
Gas Laws Practice
Quiz
•
10th - 12th Grade

10 questions
Exploring Solubility Curves
Interactive video
•
9th - 12th Grade