
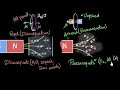
Magnetic Properties and Behavior
Interactive Video
•
Physics
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when a strong magnet is brought close to a beaker of water?
The water changes color.
The water remains unaffected.
The water repels the magnet.
The water is attracted to the magnet.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What determines whether an atom behaves like a tiny magnet?
The size of the atom.
The number of protons in the atom.
The temperature of the environment.
The presence of paired or unpaired electrons.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the phenomenon called when materials repel a magnetic field due to induction?
Electromagnetism
Paramagnetism
Diamagnetism
Ferromagnetism
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is an example of a diamagnetic material?
Aluminium
Oxygen
Copper
Iron
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What causes paramagnetic materials to become weakly attracted to magnets?
The alignment of permanent dipoles.
The presence of paired electrons.
The presence of strong magnetic fields.
The absence of thermal agitation.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does liquid oxygen exhibit stronger paramagnetism than gaseous oxygen?
It has a higher temperature.
It has a lower density.
It has more unpaired electrons.
It has a lower temperature and higher density.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does temperature affect paramagnetism?
Lower temperatures increase paramagnetism.
Lower temperatures decrease paramagnetism.
Temperature has no effect on paramagnetism.
Higher temperatures increase paramagnetism.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

11 questions
Understanding Motion: Forces and Resistance
Interactive video
•
9th - 10th Grade

8 questions
CLEAN : India's desert salt farmers feel the heat from climate change
Interactive video
•
9th - 10th Grade

8 questions
Modeling Periodic Phenomena with Trigonometric Functions
Interactive video
•
9th - 10th Grade

7 questions
Lewis Structures and Electron Pairs
Interactive video
•
9th - 10th Grade

9 questions
Hydrogen Cyanide Bonding and Properties
Interactive video
•
9th - 10th Grade

11 questions
Concept 3 - Scale, Proportion, and Quantity
Interactive video
•
9th - 10th Grade

11 questions
Cloning Processes and Techniques
Interactive video
•
9th - 10th Grade

9 questions
Magnetism and Electrical Interaction
Interactive video
•
9th - 10th Grade
Popular Resources on Wayground

7 questions
History of Valentine's Day
Interactive video
•
4th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

15 questions
Valentine's Day Trivia
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Physics

10 questions
Exit Check 3.1 - Kepler's Laws
Quiz
•
9th Grade

10 questions
Exit Check 3.2 - Force of Gavity
Quiz
•
9th Grade

27 questions
Simple Machines and Mechanical Advantage Quiz
Quiz
•
9th Grade

20 questions
Unit 8 - Energy Test - 2025-2026
Quiz
•
9th - 12th Grade

19 questions
Generators, Motors, and Transformers
Quiz
•
8th Grade - University

24 questions
IPC Thermal Energy Test Corrections
Quiz
•
9th Grade

14 questions
Bill Nye Waves
Interactive video
•
9th - 12th Grade

20 questions
Ohm's Law
Quiz
•
9th Grade