
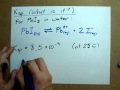
Understanding KSP and Equilibrium Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary focus of the video tutorial?
The role of temperature in solubility
The theory behind chemical equilibrium
The importance of KSP in chemical reactions
The process of dissolving solids in water
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When PbI2 dissolves in water, what ions are formed?
Lead ions and iodide ions
Lead ions and chloride ions
Sodium ions and iodide ions
Potassium ions and chloride ions
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge on the lead ion when PbI2 dissolves?
-2
-1
+2
+1
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does KQ represent in the context of the dissolution reaction?
The rate of reaction
The concentration of reactants
The concentration of products
The equilibrium constant for the reaction
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the equilibrium expression for dissolving solids, which components are ignored?
Ions and gases
Gases and solids
Solids and liquids
Liquids and gases
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is KSP considered special compared to KQ?
It measures reaction speed
It is used for liquid reactions
It is used for dissolving solids
It only applies to gases
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens if more solute is added beyond the KSP limit?
The solute dissolves completely
The excess solute precipitates as solid
The solution remains unchanged
The solution becomes unsaturated
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
