
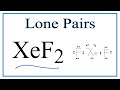
Xenon Fluoride Molecular Geometry Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Jackson Turner
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary focus when discussing lone pairs and bonding pairs in a molecule?
The lightest atom
The heaviest atom
The outermost atoms
The central atom
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many bonding pairs of electrons are present in XeF2?
One
Four
Two
Three
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is unique about xenon's electron configuration in XeF2?
It has fewer than eight electrons
It can have an expanded octet
It follows the octet rule strictly
It has no lone pairs
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many lone pairs of electrons are found in XeF2?
Two
One
Three
Four
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molecular geometry of XeF2?
Tetrahedral
Trigonal planar
Bent
Linear
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does XeF2 have a linear shape?
Due to the repulsion between three lone pairs
Because of the small size of fluorine atoms
Due to the presence of two bonding pairs
Because of the absence of lone pairs
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do lone pairs affect the shape of a molecule?
They make the molecule more compact
They do not affect the shape
They repel each other and bonding pairs
They attract bonding pairs
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
