
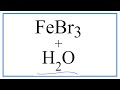
Iron(III) Bromide and Ion Behavior
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Amelia Wright
FREE Resource
Read more
8 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main purpose of examining the solubility of Iron(III) Bromide in water?
To see if it changes color in water
To determine if it can be used as a solid
To find out if it will dissolve and dissociate into ions
To check if it can be used as a gas
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which ions are we interested in when applying solubility rules to Iron(III) Bromide?
Iron(II) ion and sulfate ion
Sodium ion and bromide ion
Iron(III) ion and bromide ion
Iron(II) ion and chloride ion
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to solubility rules, how do bromide ions generally behave in water?
They are insoluble with no exceptions
They are soluble with a few exceptions
They are always insoluble
They are soluble only in hot water
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge on the bromide ion?
2+
1-
3+
0
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge on the iron ion in Iron(III) Bromide?
1+
2+
3+
4+
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many bromide ions are present when Iron(III) Bromide dissociates in water?
Three
Two
Four
One
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the term 'aqueous' indicate in the chemical equation for Iron(III) Bromide?
The substance is a solid
The substance is a gas
The substance is dissolved in water
The substance is a liquid
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
