
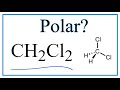
Polarity and Structure of CH2Cl2
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the polarity of dichloromethane (CH2Cl2)?
Ionic
Neutral
Polar
Non-polar
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which atoms are present in the structure of CH2Cl2?
Carbon, Oxygen, Hydrogen
Carbon, Chlorine, Hydrogen
Carbon, Fluorine, Hydrogen
Carbon, Nitrogen, Hydrogen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the electronegativity value of chlorine?
4.0
1.0
3.16
2.2
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to the rule, what electronegativity difference indicates a polar molecule?
Less than 0.5
Exactly 0.5
More than 0.5
More than 1.0
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the bond between carbon and chlorine, where do the electrons spend more time?
Around the chlorine
Equally between carbon and chlorine
In the middle of the bond
Around the carbon
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of charge does chlorine acquire in CH2Cl2 due to electron sharing?
Full negative
Partial positive
Neutral
Partial negative
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is CH2Cl2 considered a polar molecule?
It is symmetrical
It has equal distribution of charges
It is asymmetrical with distinct poles
It has no poles
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
