
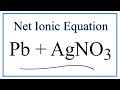
Net Ionic Equations and Spectator Ions
Interactive Video
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Hard
Aiden Montgomery
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in balancing a net ionic equation?
Determine the states of substances
Identify spectator ions
Split strong electrolytes into ions
Balance the molecular equation
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why do we put a '2' in front of silver nitrate in the molecular equation?
To balance the number of nitrate ions
To balance the number of lead atoms
To balance the number of silver atoms
To balance the number of oxygen atoms
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What state is assigned to nitrates based on solubility rules?
Gas
Aqueous
Liquid
Solid
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of a silver ion in the complete ionic equation?
1+
2-
2+
1-
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you determine the charge of lead in the products?
By the number of silver ions
By the number of oxygen atoms
By the number of nitrate ions
By the number of lead atoms
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are spectator ions?
Ions that are only in the products
Ions that participate in the reaction
Ions that do not change during the reaction
Ions that are only in the reactants
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which ions are crossed out to find the net ionic equation?
Only silver ions
Spectator ions
Only lead ions
All ions
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

6 questions
Boris Johnson Answers Press Questions at his Conservative Party Leadership Campaign Launch
Interactive video
•
10th - 12th Grade

8 questions
Meet and Greet: Bullsnake!
Interactive video
•
KG - University

6 questions
Equilibrium Constant Expression
Interactive video
•
10th Grade - University

8 questions
Practice Problem: Naming Ionic Compounds
Interactive video
•
11th Grade - University

6 questions
Why Do Atoms Bond?
Interactive video
•
11th Grade - University

8 questions
GCSE Secondary Maths Age 13-17 - Graphs: Quadratic Graphs - Explained
Interactive video
•
10th - 12th Grade

6 questions
GCSE Secondary Maths Age 13-17 - Algebra: Algebra - Explained
Interactive video
•
10th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

10 questions
Probability Practice
Quiz
•
4th Grade

15 questions
Probability on Number LIne
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

6 questions
Appropriate Chromebook Usage
Lesson
•
7th Grade

10 questions
Greek Bases tele and phon
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

20 questions
Predicting Products
Quiz
•
9th - 12th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

19 questions
Stoichiometry, % yield, Limiting Reactants
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

10 questions
Exploring Ionic and Covalent Bonding Concepts
Interactive video
•
6th - 10th Grade

7 questions
GCSE Chemistry - Balancing Chemical Equations #4
Interactive video
•
9th - 10th Grade

24 questions
Unit 2 (Part 1) Bonding Review
Quiz
•
12th Grade

12 questions
Percent Yield
Quiz
•
10th Grade