
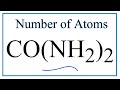
Understanding Urea and Its Composition
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Mia Campbell
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the chemical formula for urea?
CO(NH2)2
C2H4O2
CH4
NH3
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which elements are present in urea?
Carbon, Oxygen, Nitrogen, Hydrogen
Carbon, Oxygen, Sulfur, Hydrogen
Carbon, Oxygen, Nitrogen, Helium
Carbon, Nitrogen, Sulfur, Hydrogen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many nitrogen atoms are there in a single urea molecule?
3
4
2
1
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total number of atoms in a urea molecule?
8
7
6
9
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you calculate the number of carbon atoms in one mole of urea?
Multiply by 6.02 x 10^23
Divide by 6.02 x 10^23
Add 6.02 x 10^23
Subtract 6.02 x 10^23
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many oxygen atoms are there in one mole of urea?
2
4
1
3
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many nitrogen atoms are there in one mole of urea?
4
3
2
1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Popular Resources on Wayground

7 questions
History of Valentine's Day
Interactive video
•
4th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

15 questions
Valentine's Day Trivia
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

19 questions
Stoichiometry, Limiting Reactants, and Percent Yield
Quiz
•
10th Grade

10 questions
Formative 3BD: Ionic Bonds
Quiz
•
9th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

10 questions
Identifying types of reactions
Quiz
•
9th - 12th Grade

20 questions
Periodic Trends
Quiz
•
10th Grade