
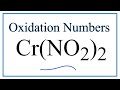
Understanding Chromium and Nitrite Ions
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Liam Anderson
FREE Resource
Read more
7 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the absence of a charge sign after chromium in chromium(II) nitrate indicate?
The compound is positively charged.
The compound is neutral.
The compound is negatively charged.
The compound is a gas.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why can chromium have different oxidation numbers?
Because it is a non-metal.
Because it is an alkali metal.
Because it is a transition metal.
Because it is a noble gas.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the ionic charge of the nitrite ion?
-1
+1
+2
0
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do the oxidation numbers of elements in an ion relate to the ion's charge?
They add up to zero.
They are always negative.
They add up to the ion's charge.
They are always positive.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What must the oxidation number of chromium be in chromium(II) nitrate for the compound to be neutral?
+3
+1
+2
+4
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the 'II' in chromium(II) nitrate?
It indicates the number of nitrate ions.
It indicates the charge of the compound.
It indicates the oxidation number of chromium.
It indicates the number of chromium atoms.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Where can you find additional help for determining the oxidation number of nitrogen in the nitrite ion?
In the video description.
In the periodic table.
In the appendix.
In the textbook.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
