
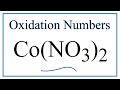
Oxidation Numbers and Cobalt Compounds
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
7 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the overall charge of cobalt(II) nitrate?
Positive
Negative
Neutral
Variable
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why can't we determine the oxidation state of cobalt just by looking at it?
Because it is a halogen
Because it is a noble gas
Because it is a transition metal
Because it is a non-metal
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the nitrate ion?
Two plus
One minus
Three minus
Zero
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do oxidation numbers in ions relate to the ion's charge?
They are always positive
They are equal to the number of atoms
They add up to the ion's charge
They are always zero
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the oxidation number of cobalt in cobalt(II) nitrate?
One plus
Two plus
Three plus
Zero
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total oxidation number of the two nitrate ions in cobalt(II) nitrate?
Plus two
Minus one
Minus two
Zero
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Where can you find additional help for finding the oxidation number of nitrogen in the nitrate ion?
In the video description
In the textbook
In the glossary
In the appendix
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
