
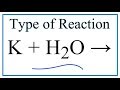
Potassium and Hydrogen Reactions
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Olivia Brooks
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of reaction occurs when potassium reacts with water?
Decomposition
Synthesis
Single displacement
Double displacement
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How can water be represented to simplify understanding of the reaction?
H2O
HOH
HHO
OH2
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a single displacement reaction, what does the metal replace?
The entire compound
The solvent
The negative ion
The positive ion
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What compound is formed when potassium reacts with water?
K2O
KOH
KH
KO2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the hydroxide ion in the reaction?
2-
2+
1+
1-
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is potassium able to replace hydrogen in the reaction?
Potassium and hydrogen have the same reactivity
Potassium is a noble gas
Potassium is more reactive than hydrogen
Potassium is less reactive than hydrogen
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Where does potassium rank in the activity series compared to hydrogen?
Above hydrogen
At the same level as hydrogen
Below hydrogen
Not in the activity series
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
