
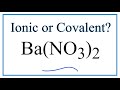
Barium Nitrate: Properties and Bonds
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Mia Campbell
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main focus of the video regarding barium nitrate?
To calculate its molecular weight
To measure its melting point
To find out if it is ionic or covalent
To determine if it is a solid or liquid
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which element in barium nitrate is a metal?
Barium
Nitrogen
Hydrogen
Oxygen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bond is formed between a metal and non-metals?
Covalent bond
Ionic bond
Metallic bond
Hydrogen bond
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the ionic charge of barium in barium nitrate?
2-
1-
2+
1+
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the nitrate ion?
2-
1-
2+
1+
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do the charges in barium nitrate balance out?
Net charge is 0
Net charge is 1-
Net charge is 1+
They don't balance
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bonds exist between nitrogen and oxygen in the nitrate ion?
Ionic bonds
Covalent bonds
Metallic bonds
Hydrogen bonds
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
