
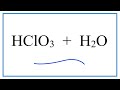
Hydronium Ion and Acid Properties
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Amelia Wright
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the nature of HClO3 when dissolved in water?
Strong base
Neutral compound
Weak acid
Strong acid
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is HClO3 considered a weak acid?
It does not dissolve in water
It forms a strong base in water
It is not listed among the strong acids
It completely dissociates in water
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to HClO3 in water?
It fully ionizes into H+ and ClO3-
It remains as HClO3
It forms a precipitate
It partially ionizes into H+ and ClO3-
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge on the hydrogen ion (H+) when HClO3 ionizes?
+2
0
+1
-1
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the state of the solution when HClO3 is in water?
Precipitated
Dynamic
Equilibrium
Static
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does 'aq' signify when written after ions?
Solid state
Gaseous state
Liquid state
Aqueous or dissolved in water
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the hydronium ion?
OH-
H3O+
ClO3-
H2O
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
