
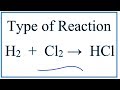
Chemical Reactions and Equations
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
6 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of reaction occurs when hydrogen gas reacts with chlorine gas to form hydrogen chloride?
Single displacement
Combination
Decomposition
Double displacement
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following best describes a synthesis reaction?
Two or more reactants combine to form a single product.
A single compound breaks down into two or more products.
An element replaces another in a compound.
Ions are exchanged between two compounds.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the state of hydrogen chloride formed in the reaction between hydrogen and chlorine gases?
Solid
Liquid
Gas
Plasma
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the reaction between hydrogen and chlorine considered exothermic?
It absorbs heat from the surroundings.
It releases a significant amount of heat.
It requires a catalyst to proceed.
It occurs only at high temperatures.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What coefficient is used to balance the hydrogen chloride in the equation H2 + Cl2 → HCl?
4
3
2
1
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a balanced chemical equation, what must be equal on both sides?
The total mass
The number of atoms for each element
The volume of gases
The number of molecules
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
