
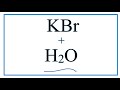
Dissociation and Properties of KBr
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
7 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of compound is potassium bromide (KBr)?
Molecular compound
Metallic compound
Ionic compound
Covalent compound
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What charge does potassium (K) have in the periodic table?
+2
-1
+1
0
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In which group of the periodic table is bromine (Br) found?
Group 2
Group 1
Group 17
Group 18
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to KBr when it is dissolved in water?
It forms a precipitate
It dissociates into ions
It remains unchanged
It evaporates
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are the ions formed when KBr dissociates in water?
K2+ and Br2-
K2+ and Br-
K- and Br+
K+ and Br-
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does 'aq' signify when written after an ion?
The ion is in solid form
The ion is in gaseous form
The ion is dissolved in water
The ion is in liquid form
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it unnecessary to write H2O on the product side of the equation?
Because water evaporates
Because 'aq' indicates the ions are dissolved in water
Because the reaction does not occur
Because water is not involved in the reaction
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
