
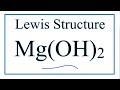
Understanding Mg(OH)2 and Hydroxide Ions
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
8 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of ion is formed by the combination of oxygen and hydrogen in Mg(OH)2?
Hydroxide ion
Carbonate ion
Nitrate ion
Sulfate ion
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the formation of Mg(OH)2, what charge does magnesium carry?
+1
+2
-1
-2
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many hydroxide ions are present in the formula Mg(OH)2?
One
Two
Four
Three
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the hydroxide ion in Mg(OH)2?
Negative
Variable
Neutral
Positive
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of using brackets in the Lewis structure of Mg(OH)2?
To indicate the number of atoms
To show the charge of ions
To represent the molecular weight
To denote the state of matter
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a formula unit in the context of Mg(OH)2?
A single molecule
A type of chemical bond
A repeating unit in a crystal
An isolated atom
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the formula unit of Mg(OH)2 help us understand?
The solubility in water
The melting point
The electron transfer process
The color of the compound
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
