
Understanding CH4 Molecular Structure and Polarity
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Ethan Morris
FREE Resource
Read more
7 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
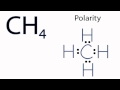
What is the initial step in determining the polarity of CH4?
Analyzing its color
Examining the Lewis structure
Measuring its temperature
Checking its solubility
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to visualize CH4 in three dimensions?
To understand the spread of hydrogen atoms
To measure its weight
To determine its color
To calculate its density
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What shape does the CH4 molecule form?
Square planar
Linear
Trigonal planar
Tetrahedral
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How are the hydrogen atoms arranged around the carbon atom in CH4?
Randomly
In a circular pattern
Equidistantly
In a straight line
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the net polarity of the CH4 molecule?
Metallic
Polar
Nonpolar
Ionic
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does CH4 have no net difference in polarity?
Because of its color
Due to its symmetrical shape
Because it is a gas
Due to its high temperature
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the symmetrical arrangement of hydrogen atoms in CH4?
It causes CH4 to be reactive
It makes CH4 a polar molecule
It makes CH4 a solid at room temperature
It results in no areas of charge imbalance
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
