
SO3 Molecular Structure and Polarity
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Aiden Montgomery
FREE Resource
Read more
6 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
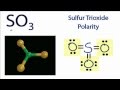
What is the bond angle in the trigonal planar geometry of SO3?
180 degrees
120 degrees
109.5 degrees
90 degrees
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is SO3 considered nonpolar based on its Lewis structure?
It has a linear shape.
The oxygens are asymmetrical.
The oxygens are symmetrical and spread out evenly.
It has a tetrahedral shape.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the shape of the SO3 molecule in three dimensions?
Linear
Bent
Trigonal planar
Tetrahedral
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the 3D shape of SO3 contribute to its nonpolar nature?
It makes the molecule linear.
It creates a positive pole.
It ensures no poles are formed.
It creates a negative pole.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the electrostatic potential of SO3 indicate about its polarity?
It has a positive side.
It has a negative side.
There are no distinct poles.
It is highly polar.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What conclusion can be drawn about the polarity of SO3 from its electronic structure?
SO3 is nonpolar because it is linear.
SO3 is nonpolar due to the absence of poles.
SO3 is polar because it has a positive and negative side.
SO3 is polar due to its asymmetrical shape.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
