
Polarity and Structure of SO2
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
6 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What aspect of the SO2 molecule's Lewis structure suggests it might be polar?
Symmetry on the top and bottom
Symmetry on the left and right
Presence of a lone pair of electrons
Absence of double bonds
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the three-dimensional structure of SO2 differ from a straight molecule?
It is bent
It is planar
It is linear
It is tetrahedral
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
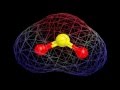
What color represents sulfur in the three-dimensional model of SO2?
Red
Blue
Green
Yellow
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What role do the valence electrons play in the polarity of SO2?
They make the molecule nonpolar
They contribute to the molecule's bent shape
They are not present in the molecule
They neutralize the molecule
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the molecular surface analysis of SO2 reveal?
The molecule is nonpolar
The molecule has no charge
The molecule has two poles
The molecule is symmetrical
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which area of the SO2 molecule is more positive according to the molecular surface analysis?
The blue area
The yellow area
The red area
The green area
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
