
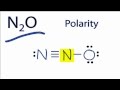
Polarity and Electronegativity of N2O
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Olivia Brooks
FREE Resource
Read more
5 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in determining if N2O is a polar molecule?
Check the molecular weight of N2O.
Examine the atoms attached to the central nitrogen.
Measure the temperature of N2O.
Determine the color of N2O.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the oxygen atom in N2O more electronegative than the nitrogen atom?
Because oxygen is a gas and nitrogen is a solid.
Because oxygen is located to the right of nitrogen on the periodic table.
Because oxygen has more protons than nitrogen.
Because oxygen is heavier than nitrogen.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What causes the formation of poles in N2O?
The presence of hydrogen bonds.
The difference in electronegativity between oxygen and nitrogen.
The equal distribution of electrons.
The symmetrical structure of N2O.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the result of having a difference in electronegativity in N2O?
N2O becomes a non-polar molecule.
N2O has a uniform charge distribution.
N2O becomes a solid at room temperature.
N2O forms a polar molecule.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is N2O considered a polar molecule?
Because it has a high boiling point.
Because it has a symmetrical structure.
Because it has a nitrogen atom on one side and an oxygen atom on the other.
Because it is a gas at room temperature.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
