
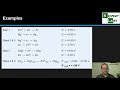
Understanding Cell Voltage and Standard Reduction Potentials
Interactive Video
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Hard

Jennifer Brown
FREE Resource
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of a more positive standard reduction potential?
It means the reaction is non-spontaneous.
It shows the reaction will not occur.
It suggests a higher likelihood of reduction.
It indicates a higher likelihood of oxidation.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which formula is used to calculate cell voltage from standard electrode potentials?
E° cell = E° anode - E° cathode
E° cell = E° cathode + E° anode
E° cell = E° cathode - E° anode
E° cell = E° anode + E° cathode
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What must be true for a reaction to be spontaneous in terms of E° cell?
E° cell must be positive.
E° cell must be zero.
E° cell must be negative.
E° cell must be less than zero.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in calculating cell voltage?
Add the half-equations together.
Multiply the half-equations.
Reverse the half-equations.
Write down the two possible reduction half-equations.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why should E° values not be multiplied when balancing equations?
They are only used for oxidation reactions.
They change with temperature.
They are innate properties and do not depend on molarity.
They depend on the number of moles.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the zinc and silver reaction example, which metal acts as the active metal?
Neither
Both
Silver
Zinc
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the calculated cell voltage for the zinc and silver reaction?
1.00 V
0.46 V
0.76 V
1.56 V
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
