
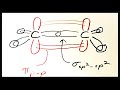
Understanding SP2 Hybridization and Isomerism
Interactive Video
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Hard

Jennifer Brown
FREE Resource
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bond is formed by the direct head-on overlap of orbitals?
Pi bond
Sigma bond
Gamma bond
Delta bond
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In SP2 hybridization, how many unhybridized p orbitals remain?
Two
Three
None
One
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the spatial arrangement of atoms in ethylene?
Tetrahedral
Pyramidal
Linear
Planar
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What phenomenon arises due to restricted rotation around a double bond?
Tautomerism
Resonance
Cis-trans isomerism
Free rotation
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which molecule is used as an example to explain free rotation in single bonds?
Butane
Ethane
Propane
Ethylene
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is required to rotate around a double bond?
Adding a catalyst
Increasing temperature
Breaking the pi bond
Breaking the sigma bond
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molecular formula of 1,2-dibromoethane?
C2H5Br2
C2H2Br2
C2H6Br2
C2H4Br2
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
